 For convenience, here is the equation again:Ħ) Plugging values into the expression gives:ħ) Two points need to be made before going on:ġ) Where did the 64.0 value come from? It was given in the problem.Ģ) Make sure to write (2x) 2 and not 2x 2. It is simply the initial conditions with the change applied to it:ĥ) We are now ready to put values into the equilibrium expression. Since we have only one equation (the equilibrium expression) we cannot have two unknowns.Ĥ) The equilibrium row should be easy. In problems such as this one, never use more than one unknown. In fact, always use the coefficients of the balanced equation as coefficients on the "x" terms. HI is being made twice as fast as either H 2 or I 2 are being used up. The positive signifies that more HI is being made as the reaction proceeds on its way to equilibrium. For every one H 2 used up, one I 2 is used up also. We know this from the coefficients of the equation. 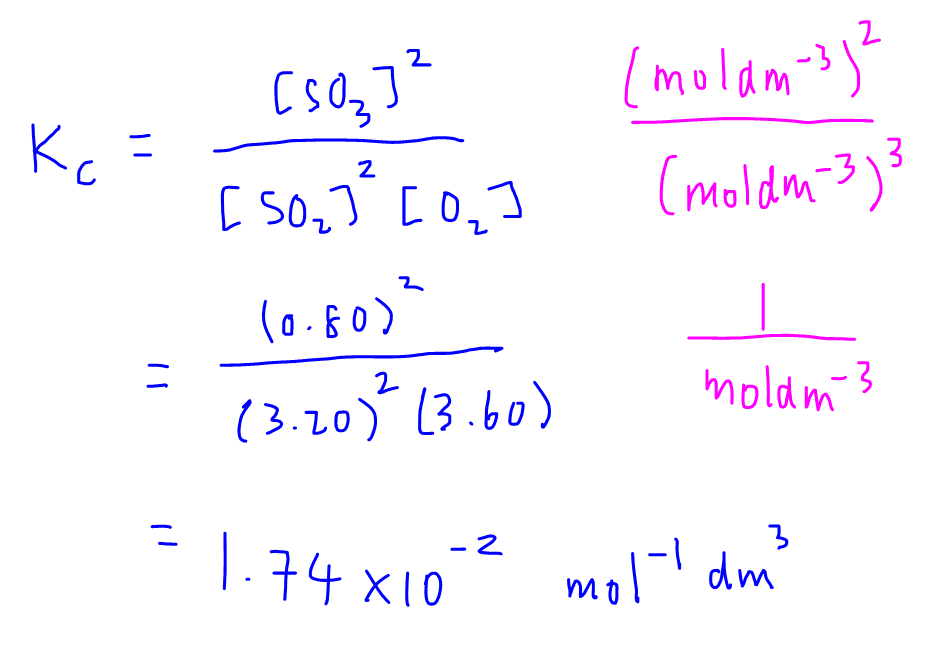
What we do know is that an EQUAL amount of each will be used up. 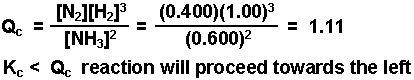
X signifies that we know some H 2 and I 2 get used up, but we don't know how much. The minus sign comes from the fact that the H 2 and I 2 amounts are going to go down as the reaction proceeds. This is the one that causes the most difficulty in understanding: The first two values were specified in the problem and the last value ( = 0) come from the fact that the reaction has not yet started, so no HI could have been produced yet.ģ) Now for the change row. The ChemTeam hopes you notice that I, C, E are the first initials of Initial, Change, and Equilibrium.Ģ) Now, let's fill in the initial row. Just in case you are not sure, the subscripted zero, as in o, means the initial concentration.Ĭalculate all three equilibrium concentrations when o = o = 0.200 M and K c = 64.0.ġ) The solution technique involves the use of what is most often called an ICEbox. In this type of problem, the K c value will be given You just plug into the equilibrium expression and solve for K c.Ĭalculating equilibrium concentrations from a set of initial concentrations takes more calculation steps. How many grams of NaNO 3 are produced when 5.3 grams of Na 2CO 3 are added to 250.0 mL of 0.ChemTeam: Calculating Equilibrium Concentrations from Initial ConcentrationsĬalculating Equilibrium Concentrations from Initial ConcentrationsĬalculating K c from a known set of equilibrium concentrations seems pretty clear. In this module, these five steps will be used to solve the following stoichiometry problem (this problem will appear on all subsequent pages): 
Step 4: Determine the Change (moles) amount for one substance in 
Step 3: If any of the known information is given as a mass or Step 2: Decide what information about the problem is known and Any stoichiometry problem can be solved by following the same series of steps:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
